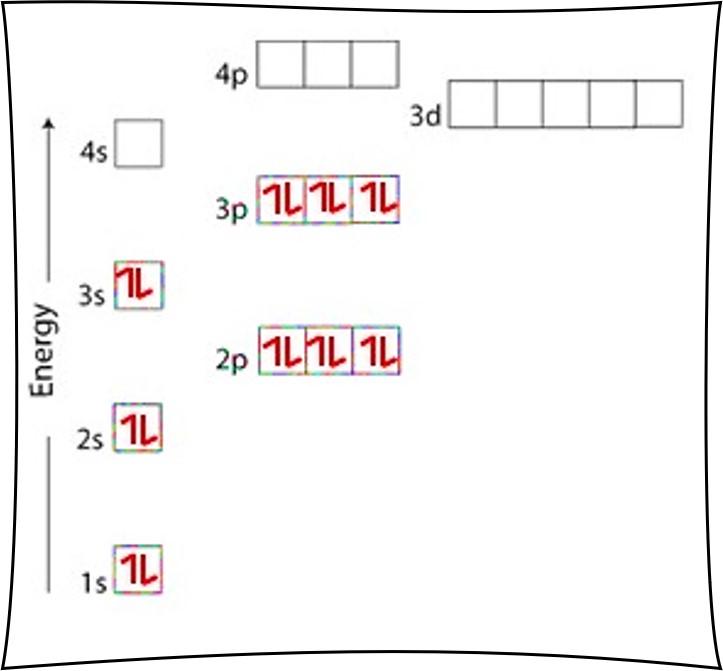
39 choose the orbital diagram that represents the ground state of n
1s 2s 2p D) 1L | 11 1 1s 2s 2p Question 4 Choose the orbital diagram that represents the ground state of N. uDENTIA Question 5 Choose the orbital diagram that represents the ground state of Neon. Question 6 Choose the orbital diagram that represents the ground state of Be. Question 7 Choose the orbital diagram that is incorrectly written?
Ans- (b) As Nitrogen contains 7 electrons in its ground state. 1st two electrons w …. View the full answer. Transcribed image text: Choose the orbital diagram that represents the ground state of N. Previous question Next question.
Question 2 05 out of 05 points choose the statement that is true. Choose the orbital diagram that represents the ground state of n. Orbital diagram that represents the ground state of n 1s2 2s2 2p3 give the set of four quantum numbers that could represent the last electron added using the aufbau principle to the cl atom.

Choose the orbital diagram that represents the ground state of n
D) E) Answer: E 15) Write out the orbital diagram that represents the ground state of As. How many unpaired electrons are there? A) 0 B) 4 C) 3 D) 2 E) 1 Answer : C
Choose the orbital diagram that represents the ground state of N. 1 The condensed electron configuration of silicon, element 14, is_ [He]2s42p6 b. [Ne]2p10 [Ne]3s23p2 d. [He]2s4 [He]2s62p2 Enthalpy is an expression for the heat content b. energy state c. reaction rate d. activation energy If AH reaction is -100 kcal/mol, it indicates the ...
Choose the orbital diagram that represents the ground state of N. Choose the valence orbital diagram that represents the ground state of Zn. Give the ground state electron configuration for Se. [Ar]4s23d104p4. Give the ground state electron configuration for I. [Kr]5s24d105p5.
Choose the orbital diagram that represents the ground state of n.
Choose the ground state electron configuration for Cr3+ [Ar]4s^2 3d^1. Choose the valence orbital diagram that represents the ground state of Sr2+ E. three 4p boxes are completely filled. Identify the isoelectronic elements. P^3-, S^2-, C^1-, K+, Ca^2+
Determine the end (final) value of n in a hydrogen atom transition, if the electron starts in n = 1 and the atom absorbs a photon of light with an energy of 2.044 * 10^-18 J 3; 4; 2; 5; 6; How many different values of l are possible in the third principal level? 1; 2; 3; 0; 4; Give the ground state electron configuration for Pb [Xe]6s2 6p2 [Xe ...
Choose the valence orbital diagram that represents the ground state of Ni. asked Jul 31, 2019 in Chemistry by kiwis. general-chemistry.
Choose the orbital diagram that represents the ground state of N. ... Which of these electron diagrams could represent the ground state of the p valence electrons of carbon? Of the following listed orbital choices - 1p, 2d, 3d, & 4f , only _____ can exist.a. 1pb. 3dc. 2d and 3dd. 3d, and 4fe. 4f ...
Exam 4 Review: Ch.8-9. Electromagnetic radiation with a wavelength of 745 nm appears as red light to the human eye. The energy of one photon of this light is ________ J. Calculate the wavelength (in nm) of the blue light emitted by a mercury lamp with a frequency of 6.19 × 10^14 Hz. Nice work!
Choose the orbital diagram that represents the ground state of N. orbital diagram where 1s and 2s orbitals contain 1 pair of electrons each. 2p orbitals are empty. orbital diagram where 1 s and 2 s orbitals contain 1 pair of electrons each. 2 p orbitals contain 3 pairs of electrons.
Choose the orbital diagram that represents the ground state of N. Learn this topic by watching The Electron Configuration Concept Videos All Chemistry Practice Problems The Electron Configuration Practice Problems
Orbital diagram that represents the ground state of n 1s2 2s2 2p3 give the set of four quantum numbers that could represent the last electron added using the aufbau principle to the cl atom. Be sure to. 5 choose the valence orbital diagram that represents the ground state of zn.
Question 4-7 refers to the following orbital diagram A) 11 1 1s 2s 2p B) 11 1L 1 1s 2s 2p C) 11 1s 2s 2p D) 1L 1 1 | 1 1s 2s 2p Question 4 Choose the orbital diagram that represents the ground state of N. Question 5 Choose the orbital diagram that represents the ground state of Neon.
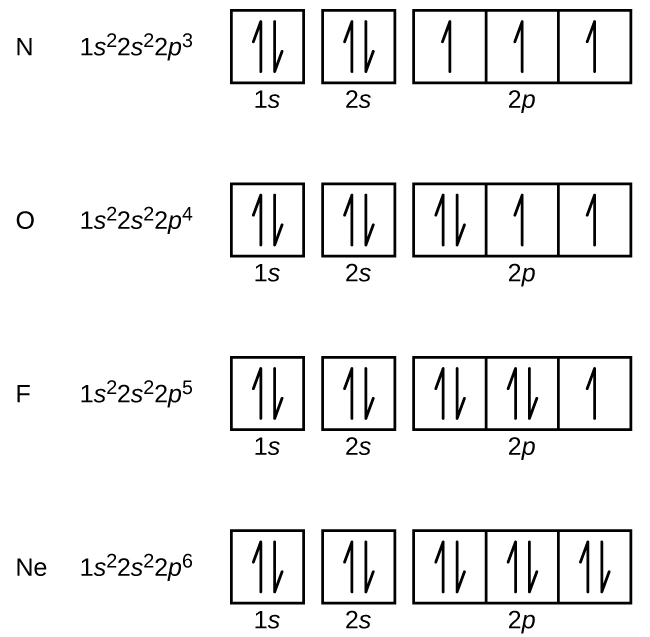
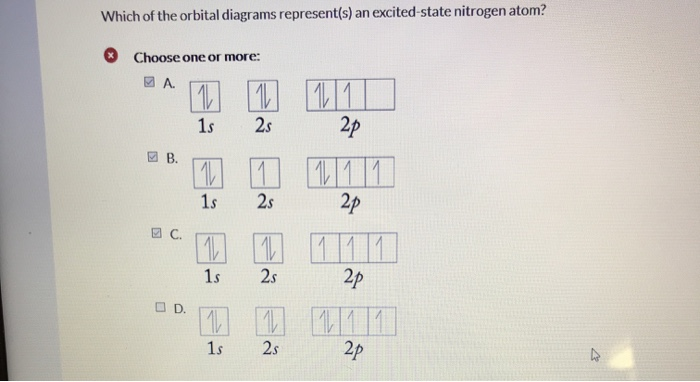
Ground or Excited State for Nitrogen. In question 1.69 (b), there is a picture which shows the electron configuration for Nitrogen. There are two arrows for the 1s orbital, 2 arrows in the 2s orbital, and one arrow in each of the three 2p orbitals. The question asks us to determine whether the electron configuration represents the excited state ...
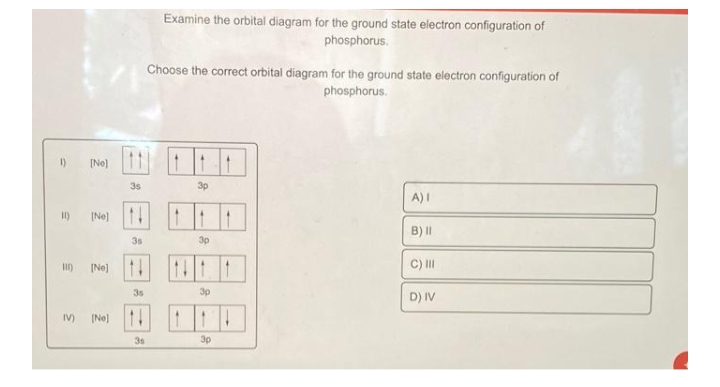
View Test Prep - Practice questions-Periodic properties-Set 3.doc from CHM 120 at University of Toronto, Mississauga. 1) Choose the orbital diagram that represents the ground state of
12) Give the set of four quantum numbers that represent the last electron added (using the Aufbau principle) to the Cl atom. A) n = 3, l =1 , ml = 0, ms = -
Choose the orbital diagram that represents the ground state of N. 1s:^> 2s:^> 2p:^^^; >=down arrow; Nitrogen has atomic # of 7. Choose the valence orbital diagram that represents the ground state of Zn. 4s:^>(4s^2) 3d:^> ^> ^> ^> ^> (3d^10) The element that corresponds to the electron configuration 1s^2 2s^2 2p^6 3s^2 3p^6 4s^1 3d^5 is _____. iron
1s 2s 2p D) 1L | 11 1 1s 2s 2p Question 4 Choose the orbital diagram that represents the ground state of N. uDENTIA Question 5 Choose the orbital diagram that represents the ground state of Neon. Question 6 Choose the orbital diagram that represents the ground state of Be. Question 7 Choose the orbital diagram that is incorrectly written?
Choose the valence orbital diagram that represents the ground state of Sr2⁺. asked Jul 31, 2019 in ... Choose the valence orbital diagram that represents the ground state of S2-asked Sep 19, 2020 in Chemistry by bonilla411. general-chemistry; Choose the valence orbital diagram that represents the ground state of Br-. asked Jul 31, 2019 in ...
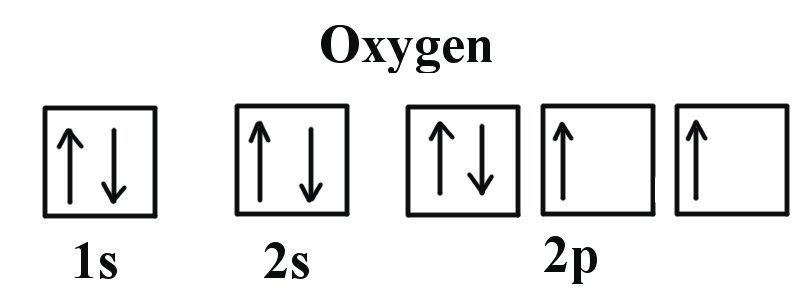
Ground State Electron Configuration For Nitrogen. When we talk about the electronic configuration, then the ground state Nitrogen Electron Configuration is written as 1s 2 2s 2 2p 3.Below you can get the full image representation which will help you to understand the topic more easily.
FREE Expert Solution. We are being asked to determine the orbital diagram that represents the ground state of N. To determine the orbital diagram, we need to know which orbital of nitrogen has its valence electrons. We will write the electron configuration of nitrogen metal. 84% (365 ratings)
Answer (1 of 4): Nickel is atomic number 28; therefore, it has 28 electrons in its orbitals. The filling rules are as follows: 1. Aufbau Principle: Lowest energy levels fill first. 2. Pauli Exclusion Principle: Only 2 electrons per orbital, they must have opposite spin. 3. Hund's Rule: Given sev...
Question 4-7 refers to the following orbital diagram A) 1 1 1 1s 2s 2p B) 1L 1L 1 1s 2s 2p C) 1s D) 2s 2p 1L | 11 | 11 1s 2s 2p Question 4 Choose the orbital diagram that represents the ground state of N. Question 5 Choose the orbital diagram that represents the ground state of Neon.
Choose the orbital diagram that represents the ground state of n. The orbital diagram for a ground state nitrogen atom is. The electron configuration of a ground state co atom is 1b a ar4s24d7 b ar4s13d5 c 1s22s22p63s23d9 d ar4s23d7 e ne3s23d7.





















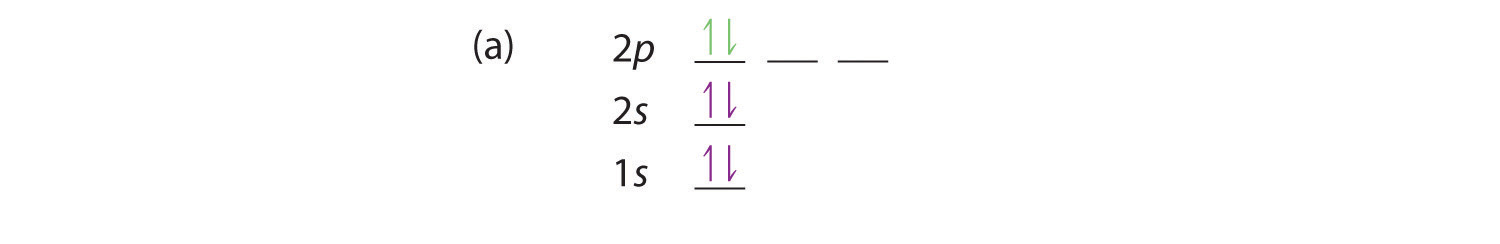






0 Response to "39 choose the orbital diagram that represents the ground state of n"
Post a Comment